
One of the most striking examples of gut plasticity can be observed in animals that are exposed to prolonged periods of fasting, such as hibernating animals or phyton snakes that goes for months without eating, where the gut shrinks with as much as 50%, but recovers in size following a few days of re-feeding. Importantly, the capacity of the gut to undergo resizing is broadly conserved. Hence, in humans, an increase in gut size is observed during pregnancy, which facilitates the uptake of nutrients to support the growth of the fetus.
The Colombani Andersen lab at the section of Cell & Neurobiology, Department of Biology, University of Copenhagen uses the fruit fly, Drosophila, to study the mechanisms that regulate gut plasticity. The results have just been published in the scientific journal Nature Communications.
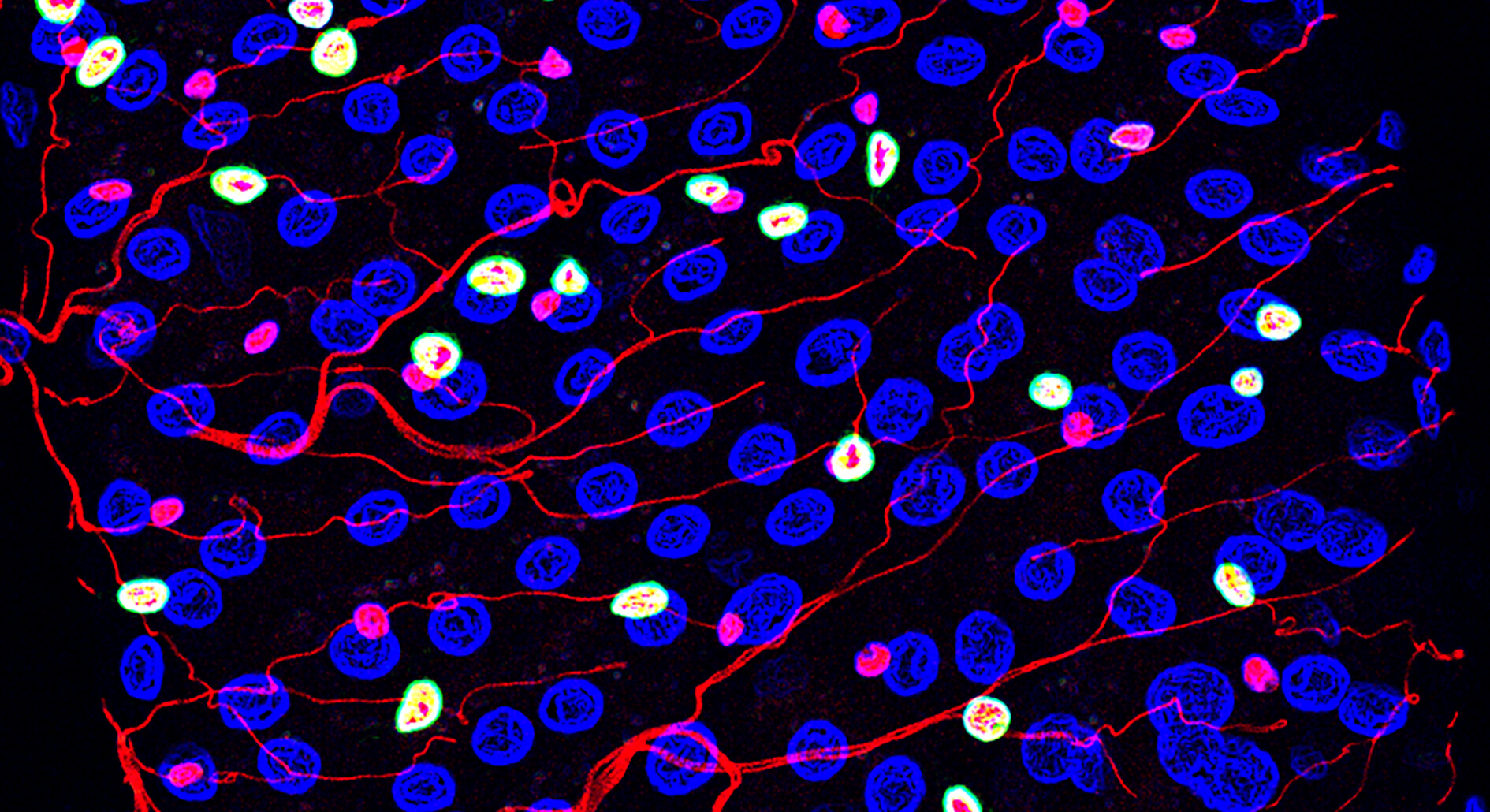
– “Taking advantage of the broad genetic toolbox available in the fruit fly, we have investigated the mechanisms underpinning nutrient-dependent gut resizing”, says Dr. Ditte S. Andersen.
The results show that nutrient deprivation results in an accumulation of progenitor cells that fail to differentiate into the mature cells causing the gut to shrink. Upon refeeding these stalled progenitor cells readily differentiate into mature cells to promote regrowth of the gut.
Ditte S. Andersen continues: – “We have identified activins as critical regulators of this process. In nutrient restrictive conditions, activin signaling is strongly repressed, while it is reactivated and required for progenitor maturation and gut resizing in response to refeeding. Activin-dependent resizing of the gut is physiologically important as inhibition of activin signaling reduces survival of flies to intermittent fasting”.
Regulators of organ plasticity are essential for host adaptation to an ever-changing environment, however, the same signals are often deregulated in cancers. Indeed, mutations affecting activin signaling are frequent in cancer cells in a variety of tissues. Our study provides a starting point for investigating the link between aberrant activin signaling and the development of colorectal cancers and sets the stage for exploring the efficiency of anti-activin therapeutic strategies in treating colorectal cancers.

