If the development of blood vessels in the placenta is impaired, fetal growth retardation may result. Scientists from the German Cancer Research Center (DKFZ) and the Mannheim Medical Faculty of Heidelberg University discovered that the correct development of functioning blood vessels in the mouse placenta is controlled epigenetically: One of the enzymes that modify gene activity using methyl groups is responsible. The researchers also observed a connection with a deficiency of this “methyltransferase” in a well-known pregnancy complication.
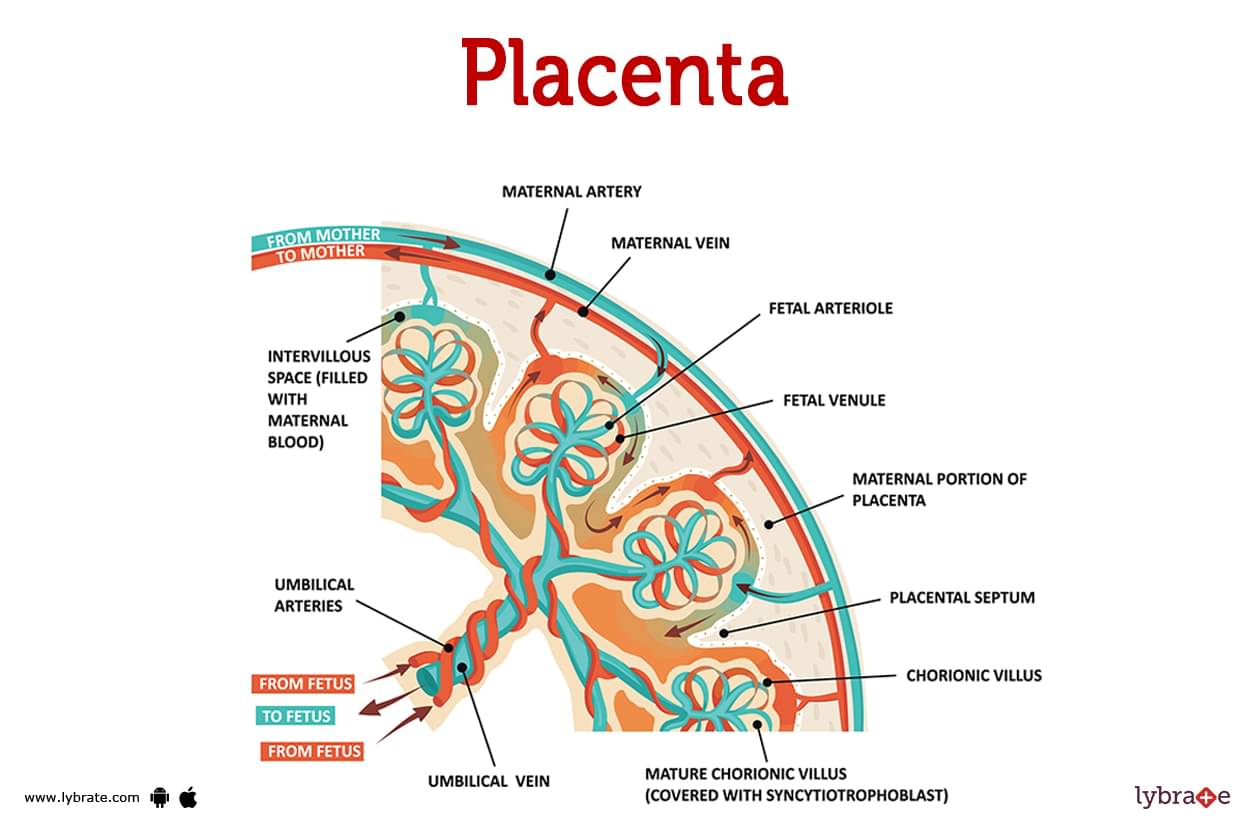

In all female mammals, including humans, the growing fetus in the uterus is supplied via the placenta. Through this temporary organ, the fetus is connected to the mother’s bloodstream, receiving nutrients and oxygen and releasing waste products.
In the case of placental insufficiency, the placenta is not supplied with a sufficient amount of blood and the exchange of substances between the placenta and fetus does not function properly. This endangers the supply of the fetus. As a rule, a developmental disorder of the blood vessels of the placenta is responsible for this.
Vascular specialist Hellmut Augustin from the DKFZ and the Mannheim Medical Faculty of Heidelberg University is well aware of the enormous importance of blood vessel development during pregnancy: “Abnormal growth of the placental blood vessels is the main cause of fetal growth retardation.” In order to gain a better understanding of how such malformations can arise, Augustin and his team have now examined the blood vessels of the mouse placenta at the single-cell level.
The researchers focused on the endothelial cells that line the inside of the blood vessels and play a crucial role in the formation of new blood vessels. Their focus was on the endothelial cells from the area of the mouse placenta that corresponds to the chorionic villi in humans.
The team found that in the normally formed placenta, the activity of certain critical genes in the endothelial cells decreases from the maternal to the fetal side. This zonation occurs in relation to the strength of the blood flow. What is the reason for this? Epigenetic mechanisms such as DNA methylation are responsible for stronger or weaker gene expression in the cell. Therefore, the researchers analyzed the enzymes responsible for DNA methylation, the so-called DNA methyltransferases. In doing so, DNA methyltransferase DNMT3A turned out to be mainly responsible for the methylation of the fetal placental endothelium.
When DNMT3A was genetically switched off in the endothelial cells of the mice, DNA methylation decreased and the spatial zonation of endothelial gene expression was lost. The development of the placental vasculature, which is crucial for the fetus, was impaired. This resulted in retarded growth, which was still noticeable after birth.
To find out whether these results obtained in mice correlate with findings in pregnant women, Augustin’s team scoured the gene databases: They compared previously published single-cell RNA sequence data from endothelial cells in healthy placentas with placentas from women suffering from preeclampsia. This complication can cause growth disorders in the baby because it is no longer properly supplied via the placenta. As expected based on the results obtained in the mouse, the placental endothelium of the preeclampsia patients had reduced DNMT3A expression.
“The combination of the compelling mouse data with the correlative patient data suggests that DNMT3A plays a crucial role in the healthy development of placental vessels – and that a deficiency of this enzyme could contribute significantly to the development of placental insufficiency,” explains Stephanie Gehrs, the first author of the publication. “A better understanding of the underlying mechanisms leading to placental insufficiency forms the basis for future approaches to better understand pregnancy disorders and possibly treat them in a more targeted manner.”
Research: The spatial zonation of the murine placental vasculature is specified by epigenetic mechanisms

